Foundayo became available in U.S. pharmacies on April 9, 2026, and I’ve already had three friends text me about it. The appeal is obvious: it’s a once-daily pill for weight loss, no injections, no food restrictions, no fasting requirements. You take it whenever you want and go about your day. Compared to everything else on the market right now, that’s a significant shift in how GLP-1 treatment works.
But most of the coverage I’ve seen treats Foundayo like it’s gender-neutral, and it’s not. The clinical trial data shows clear differences in how women experience this medication compared to men, particularly around hair loss and side effects. And then there’s the birth control interaction, which is a genuinely important safety issue that I’ve seen buried in fine print instead of headlined where it belongs.
So this is the guide I’d want to read if I were considering Foundayo. Not a generic medication overview. The stuff that matters specifically to women.
The Birth Control Interaction Is Non-Negotiable
I’m putting this first because it’s the most important thing in this article and the easiest to miss.
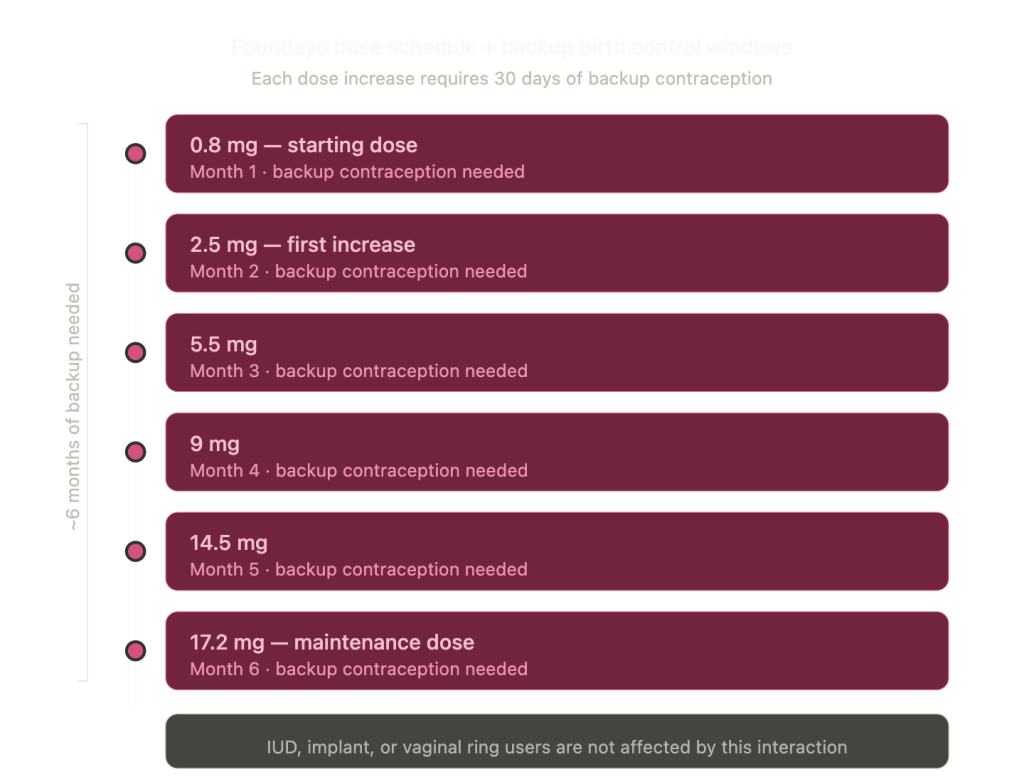
Foundayo can reduce the effectiveness of oral birth control pills. This isn’t a theoretical concern or a “talk to your doctor just in case” footnote. It’s in the official prescribing information, and the recommendation is specific: use backup contraception (condoms, IUD, vaginal ring, or another non-oral method) for 30 days after starting Foundayo and for 30 days after every dose increase.
Foundayo has six dose levels. You start at 0.8 mg and increase roughly monthly to 2.5 mg, then 5.5 mg, 9 mg, 14.5 mg, and finally 17.2 mg. That means if you’re titrating up on schedule, you could need backup birth control for the better part of your first six months on the medication. That’s not a footnote-worthy detail. That’s something you need to plan for.
If you use a non-oral method like an IUD, implant, or vaginal ring, this interaction doesn’t apply to you. But if you rely on the pill, patch, or any other oral hormonal contraceptive, this conversation with your doctor needs to happen before your first dose. Not after.
Hair Loss Hits Women Harder
Hair loss is listed as a common side effect of Foundayo. But the clinical trial data tells a more specific story than the general side effects list suggests.
In the pooled trial data, 7% of women taking Foundayo reported hair loss, compared to 0.9% of men. Even in the placebo group, women reported hair loss at a higher rate (3% vs 0.7%). The prescribing information notes that hair loss was associated with weight reduction, which makes sense. Rapid weight loss from any cause, medication, surgery, or crash dieting, can trigger telogen effluvium, a type of temporary shedding where your hair follicles shift into a resting phase.
That 7% number deserves context. It means the overwhelming majority of women won’t experience noticeable hair loss. But if you do, here’s what’s worth knowing: it’s typically temporary, it’s related to the pace of weight loss rather than the medication itself, and it usually resolves as your weight stabilizes. Maintaining adequate protein intake, not skipping meals even when your appetite is suppressed, and taking a general multivitamin can all help.
If you’re someone who’s already dealt with hair thinning, hormonal hair changes, or postpartum shedding, bring this up with your doctor proactively. You’re not being dramatic. It’s a real concern with real data behind it.
If You’re Pregnant, Planning Pregnancy, or Breastfeeding
Stop Foundayo immediately if you become pregnant. The FDA label is clear on this. Weight loss during pregnancy offers no benefit and may cause fetal harm.
There’s also a pregnancy exposure registry that Eli Lilly has established to collect data on women who took Foundayo during pregnancy, whether intentionally or not. If this applies to you, you can register by calling 1-800-545-5979. Contributing to this registry helps build the safety data that future women will rely on.
Breastfeeding is not recommended while taking Foundayo. It’s currently unknown whether orforglipron passes into breast milk, so the guidance is to avoid it entirely during nursing.
If you’re actively trying to conceive or planning to in the near future, Foundayo is probably not the right choice right now. That doesn’t mean GLP-1 treatment is off the table forever, it means the timing matters. Talk to your doctor about your timeline.
Image: Photo by RDNE
How It Compares to Oral Wegovy
This is the comparison most people are trying to make, so here’s the honest breakdown.
Oral Wegovy launched in January 2026. It contains semaglutide, the same ingredient as injectable Wegovy. It produced about 13.6% average weight loss at 64 weeks in trials. But it comes with strict dosing rules: take it first thing in the morning, on an empty stomach, with no more than 4 ounces of plain water, then wait at least 30 minutes before eating, drinking, or taking other medications. Every single day.
Foundayo showed about 12.4% average weight loss at the highest dose over 72 weeks. No food restrictions. No water restrictions. Take it whenever you want. And because it’s a small molecule (not a peptide), it doesn’t need the absorption-enhancing ingredients that make oral Wegovy so finicky.
The weight loss difference between them is real but modest: roughly 1 percentage point in favor of oral Wegovy. The convenience difference is significant. If you know yourself well enough to know that a rigid morning fasting routine will eventually lead to inconsistent dosing, Foundayo might actually produce better real-world results for you, even if the clinical numbers are slightly lower. Consistency matters more than peak efficacy.
There have been no head-to-head trials comparing Foundayo and oral Wegovy directly, so take any comparison with that context. If cost is the deciding factor, generic liraglutide is still the most affordable FDA-approved option, though less effective than both.
What It Actually Costs
Foundayo’s pricing is more transparent than most GLP-1 medications. We broke down every GLP-1 option and its real cost, which is refreshing. Here’s the breakdown:
With commercial insurance: As little as $25 per month using the Foundayo savings card, if your plan covers it. This is the best-case scenario. Call your insurance before assuming this applies to you.
Self-pay (no insurance): Starting at $149 per month for the lowest dose (0.8 mg). At the maintenance dose, expect up to $299 per month. Lilly recently matched the oral Wegovy pricing structure, so the two are comparable in cost.
Medicare Part D: Eligible beneficiaries may access Foundayo for $50 per month starting July 1, 2026.
How to access it: Foundayo is available through LillyDirect (Lilly’s direct-to-patient platform with free home delivery) telehealth providers, and U.S. retail pharmacies as of April 9, 2026.
One practical note: insurance coverage for weight-loss medications is still inconsistent. Some plans cover it, some don’t, and prior authorization requirements vary. Get a coverage check done through your doctor’s office before filling the prescription so you don’t get surprised at the pharmacy counter.
The Side Effects You’ll Actually Feel
The official list of common side effects includes nausea, constipation, diarrhea, vomiting, indigestion, stomach pain, headache, bloating, fatigue, belching, heartburn, gas, and hair loss. That reads like everything all at once, which is scary. In practice, most people experience nausea during the early weeks that gradually fades as the dose stabilizes.
The slow titration schedule (starting at 0.8 mg and increasing over months) exists specifically to minimize this. Eating smaller meals, avoiding greasy foods, and staying hydrated all help. If you’ve ever taken a medication that upset your stomach for the first week and then settled down, this is similar, just over a longer timeline because the dose keeps increasing.
Heart rate increase is another one to be aware of. Trial data showed Foundayo increased resting heart rate by 4 to 5 beats per minute on average. If you already have heart rate concerns or take medications that affect your heart, flag this with your doctor.
Serious but rare side effects include pancreatitis, gallbladder problems, kidney issues, and a thyroid tumor warning (boxed warning, the most prominent category). The thyroid concern is standard across all GLP-1 medications, not unique to Foundayo. If you have a personal or family history of medullary thyroid cancer, GLP-1 medications are not for you.
What to Bring Up at Your Appointment
Your doctor has probably seen a dozen Foundayo questions this week already. Make your appointment count by being specific:
“I take oral birth control. What’s the backup plan during titration?” Get the timeline mapped out before you leave the office.
“I’ve experienced hair thinning before. What can I do proactively?” A referral to a dermatologist or a nutritional plan focused on protein and micronutrients can start before you fill the prescription.
“Is my liver function a concern?” Foundayo is a small molecule metabolized by the liver. If you have existing liver issues or take other medications processed through the liver, this is worth discussing.
“How does this interact with my other medications?” Foundayo can affect the absorption of other oral medications. Bring a complete list, including supplements.
“What’s the plan if I decide to stop?” Weight regain after discontinuing GLP-1 medications is well-documented. Having a maintenance strategy (nutrition, exercise, potentially a lower dose) mapped out in advance makes the transition smoother.
Frequently Asked Questions
Does Foundayo affect birth control pills?
Yes. Foundayo may reduce the effectiveness of oral birth control pills. The prescribing information recommends using backup contraception (condoms, IUD, or another non-oral method) for 30 days after starting Foundayo and for 30 days after each dose increase. If you’re on a non-oral method like an IUD or implant, this interaction doesn’t apply.
How common is hair loss with Foundayo in women?
In clinical trials, 7% of women taking Foundayo reported hair loss, compared to 0.9% of men. Hair loss was associated with weight reduction rather than the medication itself. It’s typically temporary and often resolves once weight stabilizes. Maintaining adequate protein and nutrition can help minimize it.
How much does Foundayo cost per month?
With commercial insurance and a savings card: as low as $25/month. Self-pay: $149/month at the starting dose, up to $299/month at the maintenance dose. Medicare Part D: $50/month starting July 1, 2026. Coverage varies by plan, so verify before filling.
Can I take Foundayo while breastfeeding?
No. Breastfeeding is not recommended while taking Foundayo. It’s unknown whether orforglipron passes into breast milk. If you’re nursing or planning to nurse, discuss the timing of GLP-1 treatment with your doctor.
How does Foundayo compare to oral Wegovy for weight loss?
Oral Wegovy produced about 13.6% weight loss in trials; Foundayo produced about 12.4%. The key practical difference is convenience: Foundayo has no food, water, or timing restrictions, while oral Wegovy requires fasting and a 30-minute wait before eating each morning. There have been no direct head-to-head trials. Your doctor can help determine which fits your lifestyle better.
This article is for informational purposes only and is not medical advice. Foundayo is a prescription medication with potential side effects and contraindications. Always consult your doctor or healthcare provider before starting, stopping, or changing any medication.